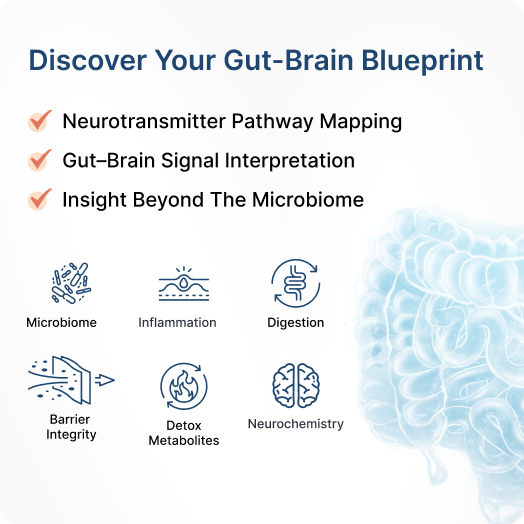
Gut Zoomer delivers a comprehensive view of how your gut and gut–brain axis are working, integrating microbiome balance, pathogens, inflammation, digestion, permeability, detox metabolites, and neurotransmitter patterns. By turning complex gut and neurochemical data into a clear, structured report, it helps you and your provider connect gut-driven contributors to symptoms and design targeted, sustainable strategies for better digestion, steadier mood and energy, and long-term resilience.
Gut microbiome imbalance and low diversity is associated with intestinal gas, autoimmune, irritable bowel syndrome, acid reflux, metabolic, cardiovascular health and inflammation.
Precision Testing
Measure diversity, richness and resilience of my gut microbiome. Test for 180+ commensal bacteria.
Personalized Solutions
Strain-specific probiotics, Prebiotic-rich foods, and postbiotics to restore microbial diversity, balance, and resilience.















.png?width=835&height=831&name=Screenshot%202025-09-05%20at%2016.11.51%201%20(1).png)
.png?width=834&height=828&name=Screenshot%202025-09-05%20at%2016.11.51%201%20(3).png)
.png?width=835&height=828&name=Screenshot%202025-09-05%20at%2016.11.51%201%20(4).png)


.png?width=457&height=457&name=Step%20Images%20(1).png)

.png?width=900&height=735&name=image%20859%20(1).png)